Introduction: A Natural Agent Bridging Disinfection and Regeneration
In regenerative endodontics, achieving effective canal disinfection without compromising stem cell viability remains one of the most significant challenges. Conventional intracanal medicaments, such as calcium hydroxide, have long served as disinfection agents but often exhibit cytotoxicity toward stem cells critical for pulp-dentin regeneration. This creates a clinical paradox: while the canal must be sterilized to prevent reinfection, the same antimicrobial agents may hinder true tissue regeneration.
Recent research has identified Benzyl Isothiocyanate (BITC), a naturally occurring compound extracted from the Miswak (Salvadora persica) plant, as a promising solution to this dilemma. A study conducted at King Saud University and published in Frontiers in Cellular and Infection Microbiology revealed that BITC can effectively eradicate key endodontic pathogens while maintaining the integrity and viability of human stem cells, thereby positioning it as a strong candidate for regenerative endodontic applications.
Understanding BITC: A Bioactive Compound from Miswak
Benzyl Isothiocyanate (BITC) is a sulfur-containing phytochemical present in several cruciferous and medicinal plants, including Miswak, papaya seeds, and garden cress. It is released when plant tissues are disrupted, serving as a natural defense molecule with potent antimicrobial and anti-biofilm capabilities. Historically, Miswak has been used for oral hygiene, and modern research continues to validate its broad-spectrum antibacterial activity.
The antimicrobial mechanisms of BITC include:
- Cell Membrane Disruption: BITC penetrates bacterial membranes, increasing permeability and inducing cell death.
- Oxidative Stress Induction: It promotes intracellular accumulation of reactive oxygen species (ROS), leading to oxidative damage.
- Virulence Inhibition: BITC downregulates bacterial virulence genes involved in adhesion and biofilm formation.
- Biofilm Degradation: It breaks down the extracellular polymeric matrix, destabilizing bacterial colonies.
What makes BITC especially noteworthy in regenerative endodontics is its selective cytotoxicity—the ability to target pathogenic cells without harming host stem cells, a property also noted in oncological research.
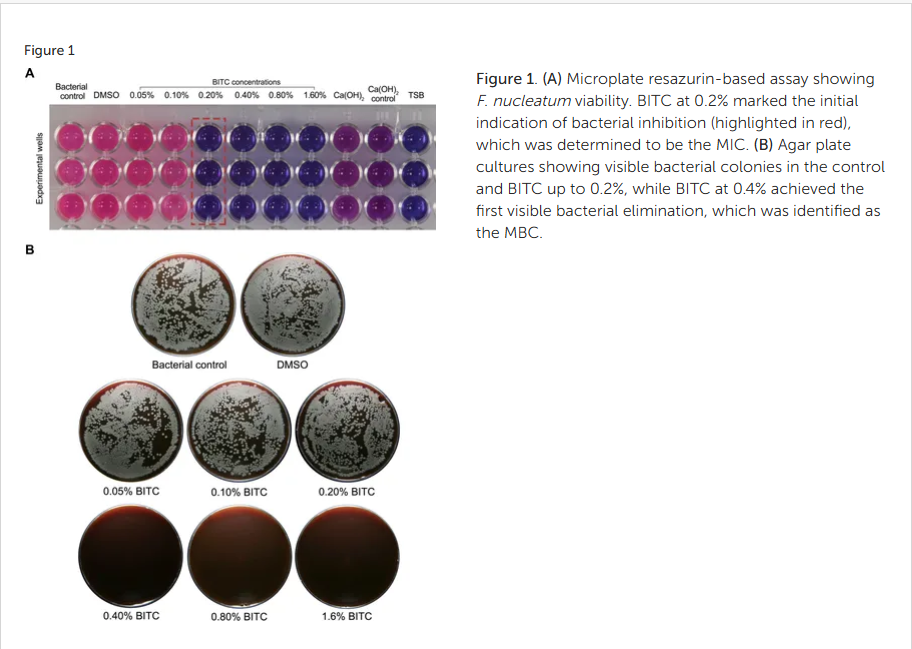
Figure 1. (A) Microplate resazurin-based assay showing F. nucleatum viability. BITC at 0.2% marked the initial indication of bacterial inhibition (highlighted in red), which was determined to be the MIC. (B) Agar plate cultures showing visible bacterial colonies in the control and BITC up to 0.2%, while BITC at 0.4% achieved the first visible bacterial elimination, which was identified as the MBC.
BITC’s Dual Role in Endodontic Regeneration
The study aimed to investigate two main aspects of BITC’s performance:
- It has antimicrobial and antibiofilm activity against Fusobacterium nucleatum, a persistent pathogen implicated in endodontic infections.
- Its biocompatibility and impact on human bone marrow-derived mesenchymal stem cells (hBMMSCs), essential for tissue regeneration within the pulp-dentin complex.
BITC’s effects were directly compared with calcium hydroxide (Ca(OH)₂), the conventional intracanal medicament used in regenerative procedures.
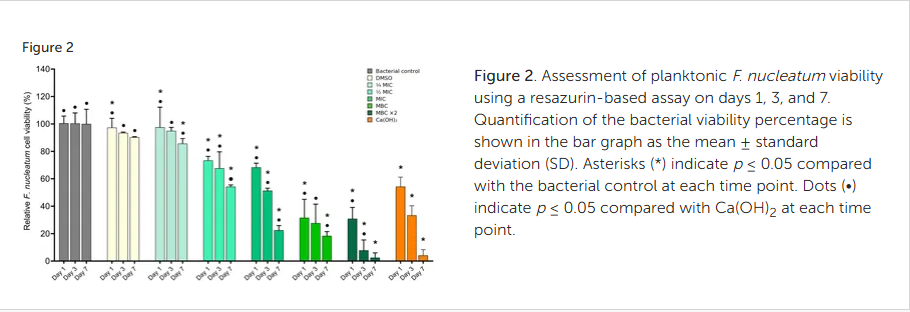
Figure 2. Assessment of planktonic F. nucleatum viability using a resazurin-based assay on days 1, 3, and 7. Quantification of the bacterial viability percentage is shown in the bar graph as the mean ± standard deviation (SD). Asterisks (*) indicate p ≤ 0.05 compared with the bacterial control at each time point. Dots (•) indicate p ≤ 0.05 compared with Ca(OH)2 at each time point.
Experimental Overview and Key Methodologies
The research utilized a combination of microbiological and cytocompatibility assays to determine BITC’s therapeutic potential:
- MIC and MBC Assessment: Established the minimum inhibitory and bactericidal concentrations of BITC.
- Biofilm Quantification: Employed crystal violet assays and confocal microscopy with live/dead staining to evaluate biofilm mass and bacterial viability.
- Surface Morphology: Scanning Electron Microscopy (SEM) was employed to visualize the structural damage induced by BITC in bacteria.
- ROS Measurement: Determined oxidative stress as a mechanism of antibacterial activity.
- Stem Cell Viability Testing: Conducted Alamar Blue assays to assess metabolic activity and cellular morphology of hBMMSCs exposed to varying BITC concentrations.
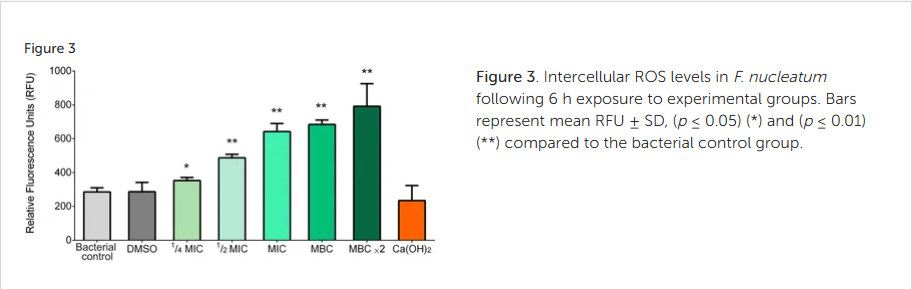
Figure 3. Intercellular ROS levels in F. nucleatum following 6 h exposure to experimental groups. Bars represent mean RFU ± SD, (p ≤ 0.05) (*) and (p ≤ 0.01) (**) compared to the bacterial control group.
Results: BITC’s Superior Performance Over Calcium Hydroxide
1. Potent Antibacterial and Anti-Biofilm Activity
BITC exhibited a remarkable bactericidal effect against F. nucleatum with an MIC of 0.2% and an MBC of 0.4%. Unlike calcium hydroxide—which required several days to achieve significant bacterial reduction—BITC achieved strong antibacterial activity within 24–72 hours. Confocal imaging confirmed extensive bacterial death and substantial biofilm disruption, with visible extracellular matrix breakdown observed via SEM analysis.
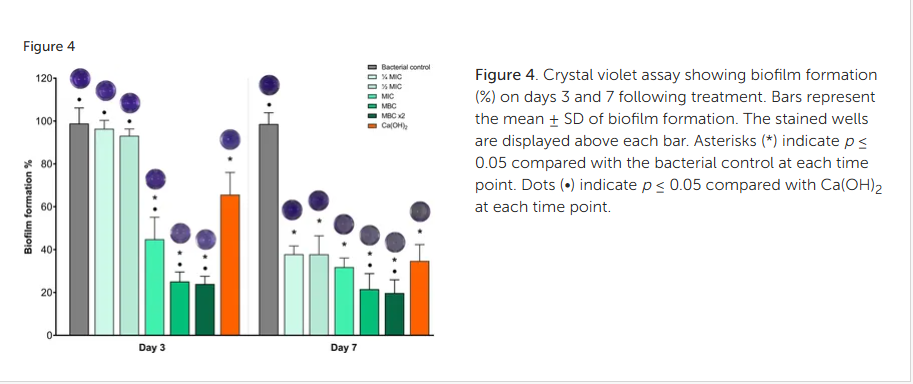
Figure 4. Crystal violet assay showing biofilm formation (%) on days 3 and 7 following treatment. Bars represent the mean ± SD of biofilm formation. The stained wells are displayed above each bar. Asterisks (*) indicate p ≤ 0.05 compared with the bacterial control at each time point. Dots (•) indicate p ≤ 0.05 compared with Ca(OH)2 at each time point.
2. ROS-Mediated Mechanism of Action
BITC induced a significant increase in intracellular ROS levels, suggesting oxidative stress as its primary antimicrobial mechanism. This contrasts with calcium hydroxide, whose effect relies on high alkalinity rather than oxidative disruption. The oxidative pathway not only promotes bacterial cell death but also minimizes the development of resistant bacterial phenotypes.
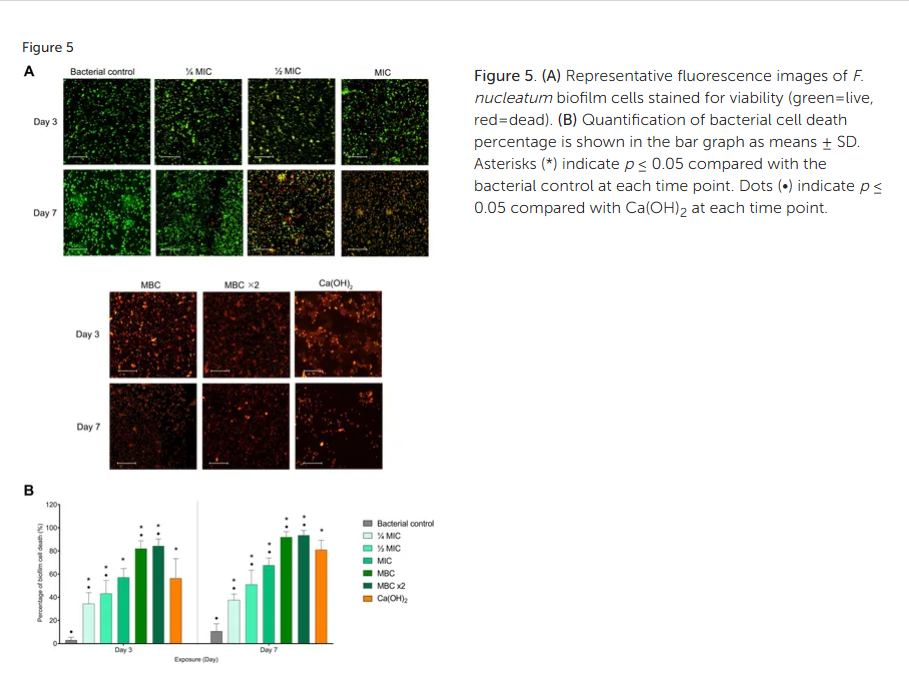
Figure 5. (A) Representative fluorescence images of F. nucleatum biofilm cells stained for viability (green=live, red=dead). (B) Quantification of bacterial cell death percentage is shown in the bar graph as means ± SD. Asterisks (*) indicate p ≤ 0.05 compared with the bacterial control at each time point. Dots (•) indicate p ≤ 0.05 compared with Ca(OH)2 at each time point.
3. High Biocompatibility with Mesenchymal Stem Cells
BITC demonstrated excellent cytocompatibility across multiple concentrations. At 0.05–0.2%, stem cell viability remained above 90%, with slight proliferative effects noted at lower doses. Even at the bactericidal concentration of 0.4%, viability remained above the acceptable 70% cytotoxicity threshold—indicating a therapeutic window suitable for clinical use.
In contrast, calcium hydroxide showed marked cytotoxicity, causing cell shrinkage, detachment, and decreased metabolic activity. Microscopic evaluation confirmed that BITC-treated stem cells preserved their normal spindle-shaped morphology, while Ca(OH)₂-treated cells exhibited stress-related alterations.
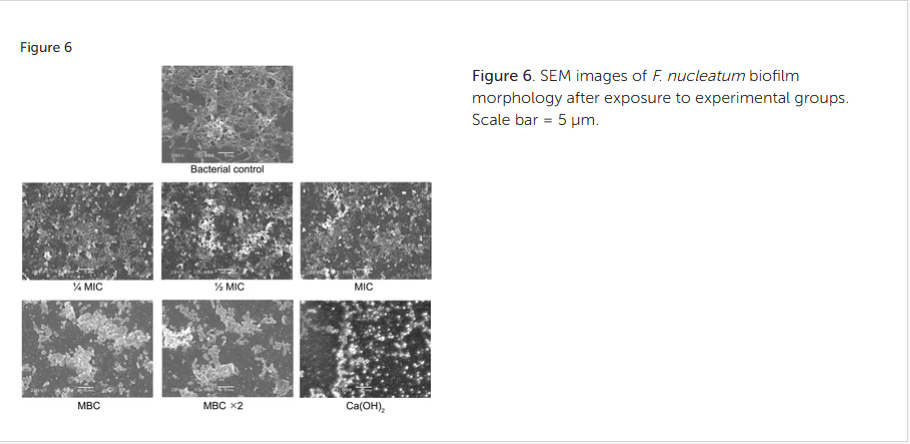
Figure 6. SEM images of F. nucleatum biofilm morphology after exposure to experimental groups. Scale bar = 5 μm.
Clinical Implications for Regenerative Endodontics
The findings provide compelling evidence that Miswak-derived BITC could address one of the key limitations in regenerative endodontic procedures—the incompatibility between effective canal disinfection and stem cell preservation. By offering rapid bactericidal effects against F. nucleatum biofilms while maintaining stem cell integrity, BITC effectively bridges this therapeutic gap.
This balance is crucial for achieving predictable outcomes in Regenerative Endodontic Procedures (REPs). Damaged or depleted stem cell populations compromise the regenerative potential of the pulp-dentin complex. Therefore, a compound like BITC, with both antimicrobial and biocompatible properties, holds strong translational promise for future clinical use.
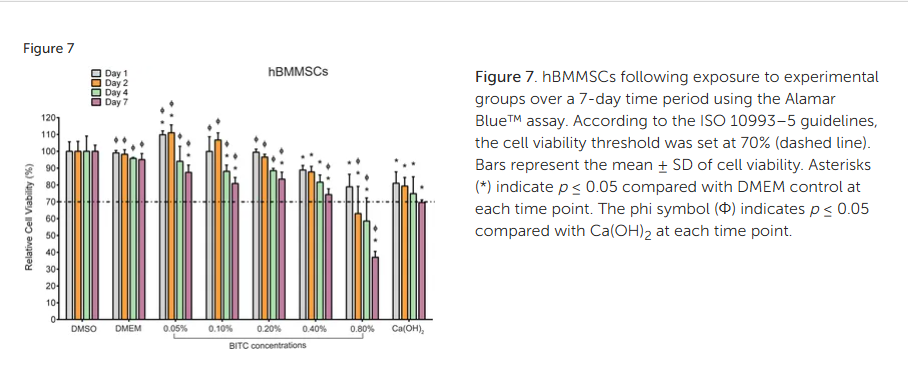
Figure 7. hBMMSCs following exposure to experimental groups over 7 days using the Alamar Blue™ assay. According to the ISO 10993–5 guidelines, the cell viability threshold was set at 70% (dashed line). Bars represent the mean ± SD of cell viability. Asterisks (*) indicate p ≤ 0.05 compared with DMEM control at each time point. The phi symbol (Φ) indicates p ≤ 0.05 compared with Ca(OH)2 at each time point.
Limitations and Future Research Directions
While the current findings are promising, further research is essential to validate BITC’s potential in vivo and under clinical conditions:
- Multi-Species Biofilm Testing: Future studies should evaluate BITC’s effect on polymicrobial endodontic biofilms to mimic clinical infection complexity.
- Stem Cell Differentiation Studies: Long-term assessments are needed to ensure BITC does not interfere with odontogenic differentiation, migration, or mineralization processes.
- Animal and Clinical Trials: Preclinical and human trials will be critical to confirm safety, efficacy, and delivery system optimization for clinical use.
- Formulation Development: Encapsulation or sustained-release systems could enhance BITC stability and controlled delivery within the root canal environment.
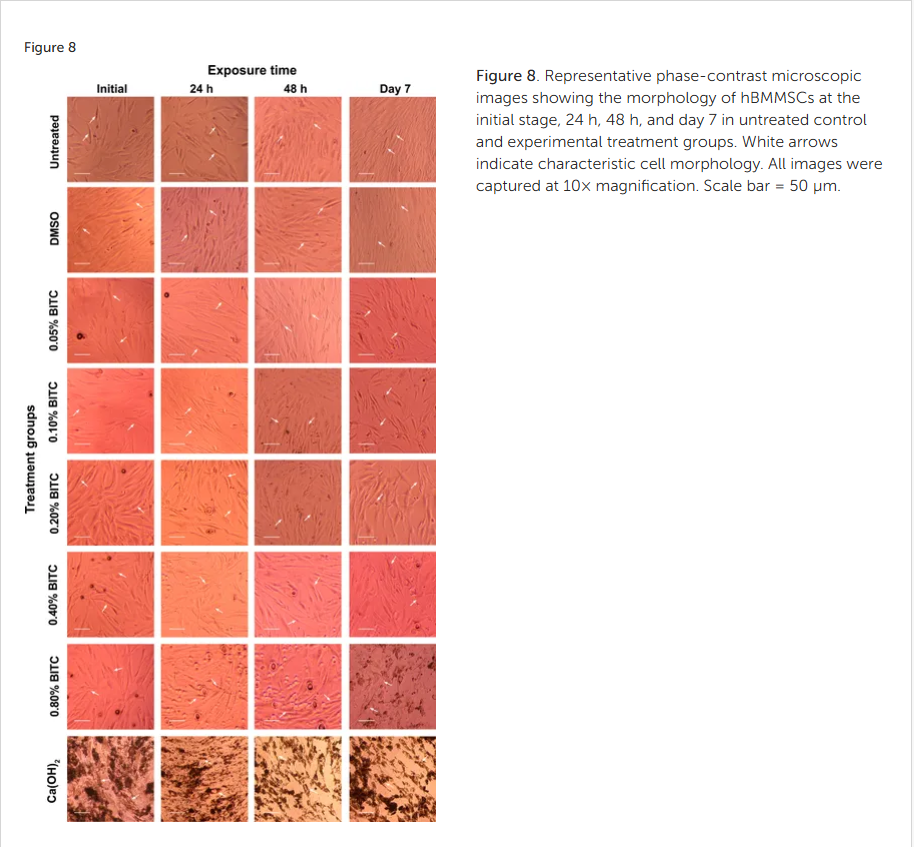
Figure 8. Representative phase-contrast microscopic images showing the morphology of hBMMSCs at the initial stage, 24 h, 48 h, and day 7 in untreated control and experimental treatment groups. White arrows indicate characteristic cell morphology. All images were captured at 10× magnification. Scale bar = 50 µm.
Conclusion: Toward a Biocompatible Future in Endodontic Regeneration
The introduction of Benzyl Isothiocyanate (BITC) from Miswak represents a significant advance in regenerative endodontics. Its ability to eradicate resistant endodontic pathogens like Fusobacterium nucleatum while preserving stem cell viability offers a paradigm shift in intracanal medicament development. Natural, biocompatible, and mechanistically distinct from traditional agents, BITC holds the potential to transform regenerative endodontic protocols into more predictable, biologically driven procedures.
As ongoing research continues to explore its translational applications, BITC may soon play a central role in next-generation endodontic therapeutics—where infection control and tissue regeneration coexist harmoniously.
Source :
Benzyl isothiocyanate exhibits antibacterial and antibiofilm activity against Fusobacterium nucleatum while preserving viability of human bone marrow mesenchymal stem cells: an in vitro study
The Role of Miswak BITC Compound in Endodontic Regeneration
Share on:
